Case Report
Redo Mitral Valve Surgery Via a Right Thoracotomy with Heart-Lung Machine: Presentation of Two Cases
Deser SB* and Demirag KM
Department of Cardiovascular Surgery, 19 Mayis University, Turkey
*Corresponding author: Serkan Burc Deser, Department of Cardiovascular Surgery, 19 Mayis University, Faculty of Medicine, 55139 Samsun, Turkey
Published: 08 Jun, 2016
Cite this article as: Deser SB, Demirag KM. Redo Mitral
Valve Surgery Via a Right Thoracotomy
with Heart-Lung Machine: Presentation
of Two Cases. Clin Surg. 2016; 1: 1024.
Abstract
The rate of redo surgeries are increasing on account of previously performed coronary artery bypass graft surgeries, aortic/ mitral valve replacement surgeries due to valve stenosis/ regurgitation,
mitral valve repair for ischemic mitral regurgitation, the aging in the population, mechanical
valve thrombosis and wearing out of the mechanical valves.Resternotomy, anterolateral right
thoracotomy or anterolateral left thoracotomy can be prefered for redo mitral valve surgery. Redo
on pump beating heart mitral valve surgery via a anterolateral right thoracotomy is safer and reduces
the injury risk of patent grafts, adherent tissues and ventricles. Here we present redo mitral valve
replacement of the two patients via anterolateral right thoracotomy
Keywords: Reoperation; Mitral valve; Thoracotomy
Introduction
The rate of redo surgeries are increasing on account of previously performed coronary artery by pass graft surgeries, aortic/ mitral valve replacement surgeries due to valve stenosis/ regurgitation, mitral valve repair for ischemic mitral regurgitation, the aging in the population, mechanical valve thrombosis and wearing out of the mechanical valves. Mitral valve surgery after previous open heart surgeryis a surgical challange.That may lead to two-threefold increase in the mortality and morbidity rather than primary surgery. Resternotomy, anterolateral right thoracotomy or anterolateral left thoracotomy can be prefered for redo mitral valve surgery. On pump standard median sternotomy approach may lead to injury of the bypass grafts or cardiac structures. History of previous mediastinitis and sternotomies, CABG with functioning grafts and previous aortic valve replacement (AVR) are the indications of thoracotomy approach. Redo on pump beating heart mitral valve surgery via a anterolateral right thoracotomy is safer and reduces the injury risk of patent grafts, adherent tissues and ventricles and also reduces the operation time, reduces amount of blood loss and improve the hemodynamics [1]. However, right pleural dissection in a redo surgery is troublesome and a surgical challenge. Injury of patent CABGsuch as internal mammarian artery (IMA), can be fatal during reoperation via resternotomy
Case Presentation
case 1
A 30 years old female with dyspnea(New York Heart Association Functional Class II-III)
on exertion and had a history of three times mitral valve replacement surgery, was admitted.
Previous mitral valve replacement surgeries were performed 4, 6 and 8 years earlier, respectively.
Transthoracic echocardiography revealed modarate mitral valve regurgitation, huge trombus on the
atrial site of the valve. Cardiac catheterization showed patent coronary arteries. European System
for Cardiac Operative Risk Evaluation II was 4.05%. Routine preparation for surgery was made
and informed consent was taken. External defibrillation pads were placed on the left anterior and
posterior chest wall before the incision was made. Under general anesthesia with double lumen
intubation, the patient was positioned with the right chest 300 elevated. A anterolateral right
thoracotomy was performed through the fourth intercostal space. Cardiopulmonary bypass was
initiated via right femoral artery and standard two stage venous cannulation was performed. The
patient’s body temperature was lowered to 250oC. Ascending aortic root cannula was placed for
deairing. Cardiopulmonary bypass was performed without aortic cross-clamping which is known
as the ‘no-touch technique’ due to severe adhesions [2]. The mean arterial perfusion pressure was
maintained at over 40 mmHg during surgery in order to keep the aortic valve closed. The patient was
kept in the Trendelenburg (head-down tilt) position and continuous aortic root venting was used to prevent air embolism. A longitudinal left atriotomy incision was
performed. Tracheoesophageal echocardiography (TEE) monitoring
was performed during surgery due to the thrombus debris on
it, previously placed mechanical heart valve was almost stucked.
Prosthetic mitral valve was replaced with a 27 mm mechanical valve
(St. Jude Medical Inc. St Paul, MN, USA) (Figure 1). The patient
was weaned from cardiopulmonary bypass after rewarming. On
post operative 43. day, respiratory infection and progressive clinical
deterioration was developed that led to sepsis despite intensive
antibiotic therapy, the patient progressively deteriorated and died.
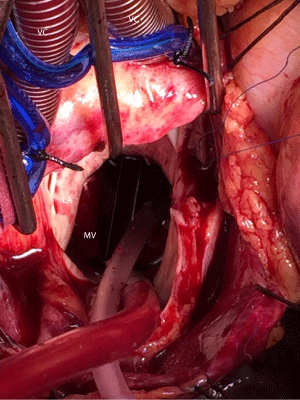
Figure 1
Figure 1
Peroperative view of the thrombosed previous mitral valve on the
atrial site. (VC: Vena cava, RA: Right atrium, MV: Mitral Valve, AV: Aortic oot
Vent, LA: Left Atrium, S: Sucker, AsA: Ascending Aorta).
case 2
A 70 years old female presented with dyspnea on exertion (New
York Heart Association Functional Class II-III) and had previously
underwent coronary artery bypass graft (CABG) x2 surgery 4 years
ago, was admitted. Cardiac catheterization showed patent bypass
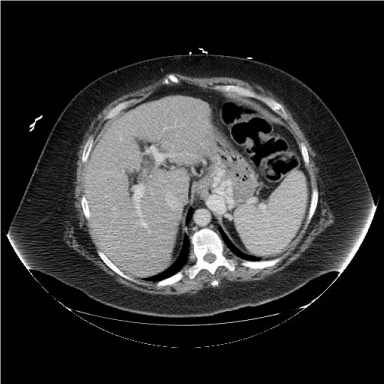
grafts. Computed tomography (CT) scan of the chest showed left
internal mammarian artery (LIMA) graft courses beneathth sternum.
Transthoracic echocardiography revealed severe mitral valve and
moderate tricuspid valve regurgitation and the ejection fraction was
47%. European System for Cardiac Operative Risk Evaluation II
was 7%. External defibrillation pads were placed on the left anterior
and posterior chest wall before the incision was made. Under
general anesthesia with double lumen intubation, the patient was
positioned with the right chest 30º elevated. The right lung deflation
was achieved. A anterolateral right thoracotomy was performed
through the fourth intercostal space (Figure 2). Cardiopulmonary
bypass was initiated via right femoral artery and standard two stage
venous cannulation was performed. TEE monitoring was performed
during surgery. Prosthetic mitral valve was replaced with a 29 mm
mechanical valve (St. Jude Medical Inc, St Paul, MN, USA) (Figure 2).
The patient was kept in the Trendelenburg (head-down tilt) position
and continuous aortic root venting was used to prevent air embolism.
Patient was weaned from cardiopulmonary bypass after rewarming.
The operation was completed without any injury on the patent’s grafts or cardiac structures and the blood loss was minimal. The heart
was not fibrillated and neither aortic cross-clamping nor cardioplegia
was required. The patient was discharged on post operative 5 day
uneventfully.
Figure 2
Discussion
Mitral valve surgery in the presence of previous sternotomy is a surgical challenge. Redo procedures are technically more complex than primary operations. Lower mortality rate can be achieved with ‘no touch technique’ in high-risked patients with lower morbidity. During standard sternotomy technique, dissection may lead to injury of the cardiac structures [3]. Avoiding of excessive aortic manipulation and dissection reduces the risk of embolisation. Thoracotomy approach have more advantages rather than a redo sternotomy for LIMA graft that lies beneath the sternum. Retrograde arterial perfusion via femoral artery cannulation reduces the risk of adverse neurologic event. On pump beating heart surgery is superior than the hypothermic fibrillatory arrest and provides sufficient myocardial perfusion during diastole and has a threefold lower risks of stroke [4]. Also cardioplegia leads to interruption of the LIMA grafts. The right thoracotomy approach provides good exposure of the mitral valve if there is not any previous pleural adhesions [5]. Trendelenburg position, continuous aortic root venting and TEE are essential to avoid air embolism. The redo on pump beating heart mitral valve surgery provides a complete myocardial perfusion during diastole which maintaines sufficient coronary perfusion to reduce myocardial ischemia and reperfusion injury [6]. Contraindications are including aortic regurgitation, right thoracic pleurodesis, low ejection fraction, multiple previous right thoracotomies, large right chest wall arteriovenous (A-V) malformation and previous right pneumonectomy [7]. In conclusion, we believe that if aortic cross-clamping can not be performed for redo surgeries, on pump beating heart mitral valve surgery via a right thoracotomy can be safely performed compared to standard sternotomy.
References
- Petracek MR, Leacche M, Solenkova N, Hoff SJ, Absi TS, Ball Sk, et al.Minimally invasive mitral valve surgery expands the surgical options for high-risks patients. Ann Surg. 2011; 254: 606-611.
- Kitamura T, Stuklis RG, Edwards J. Redo mitral valve operation via right minithoracotomy: “no touch” technique. Int Heart J. 2011; 52: 107-109.
- Kim HR, Kim GS, Yoo JK, Lee JW. Minimally Invasive Approach for Redo Mitral Valve Replacement: No Aortic Cross-Clamping and No Cardioplegia. Korean J Thorac Cardiovasc Surg. 2015; 48: 126-128.
- Gammie JS, Zhao Y, Peterson ED, O’Brien SM, Rankin JS, Griffith BP. J. Maxwell Chamberlain Memorial Paper for adult cardiac surgery. Lessinvasive mitral valve operations: trends and outcomes from the Society of Thoracic Surgeons Adult Cardiac Surgery Database. Ann Thorac Surg. 2010; 90: 1401–1410.
- Petracek MR, Leacche M, Solenkova N, Umakanthan R, Ahmad RM, Ball SK et al. Minimally Invasive Mitral Valve Surgery Expands the Surgical Options for High-Risks Patients. Ann Surg. 2011; 254: 606-611.
- Uehara K, Minakata K, Funamoto M, Yamazaki K, Marui A, Sakata R. Alternative redo sternotomy in a patient with tracheostoma and patent grafts. Asian Cardiovascular & Thoracic Annals. 2014; 22: 852–854.
- Parlar AI, Cicekcioglu F, Irdem AK, Hijazi A, Altinay L, Katircioglu SF. Mitral Valve Replacement with Beating Heart Technique via Right Thoracotomy in a Patient with Previous Bentall Operation. Thorac Cardiov Surg. 2010; 58: 246–248.