Case Report
A Case of Takotsubo Cardiomyopathy Possibly Induced by Cilostazol Administration
Fukata M1*, Hatakeyama K1,Yokoyama T1, Nakamura H1,Yasuda S1,Muta T1,Odashiro K1,MaruyamaT1,2 and Akashi K1
1Department of Medicine and Biosystemic Science, Kyushu University Faculty of Medicine, Japan
2Division for Student Counseling and Guidance, Faculty of Arts and Science, Japan
*Corresponding author: Mitsuhiro Fukata, Department of Medicine and Biosystemic Science, Kyushu University Faculty of Medicine, 3-1-1 Maidashi, Higashi-ku, Fukuoka 812-8582, Japan
Published: 05 May 2016
Cite this article as: Fukata M, Hatakeyama K, Yokoyama T,
Nakamura H, Yasuda S, Muta T. A Case
of Takotsubo Cardiomyopathy Possibly
Induced by Cilostazol Administration.
Clin Surg. 2016; 1: 1010.
Abstract
Takotsubo cardiomyopathy is a transient cardiac syndrome that involves left ventricular apical
akinesis typically triggered by a significant emotional or physical stress. Here, we report a patient
who presented with strong chest discomfort with left ventricular apical akinesis following cilostazol
administration and was diagnosed with Takotsubo cardiomyopathy. Sudden onset of the disease
immediately after cilostazol administration and no preceding emotional or physical stressors
suggested that the pharmacological activity of cilostazol had triggered Takotsubo cardiomyopathy.
Following the discontinuation of cilostazol and heart failure treatment, left ventricular dysfunction
had fully recovered.
Keywords: Takotsubo cardiomyopathy; Cilostazol; Cyclic AMP; Left ventricular obstruction
Introduction
Takotsubo cardiomyopathy is characterized by transient and regional left ventricular dysfunction, which does not correspond anatomically to coronary distribution, without obstructive coronary lesions. The onset of the disease frequently follows episodes of emotional or physiological stress. However, the preceding stressor is not clearly identifiable in part of the patients [1,2]. In the patients without preceding emotional or physiological stressors, several drugs are reported as the possible triggers [3]. We present here a case in which preceding cilostazol administration and underlying left ventricular outflow tract obstruction (LVOTO) were noted to precipitate Takotsubo cardiomyopathy.
Case Presentation
An 81-year-old woman presented with strong chest discomfort to our hospital on October 28,
2013. A physical examination revealed both an ejection systolic murmur (Levine II/VI, max at the
third intercostal space along the left sternal border) and regurgitant systolic murmur (Levine III/
VI, max at the apex). The electrocardiogram exhibited an elevated ST segment in leads II, III, aVF,
and V2-5 and negative T wave in leads I, aVL, and V4-6 (Figure 1A). The echocardiography showed
apical akinesis and basal hyper kinesis, LVOTO (pressure gradient = 55 mmHg), and severe mitral
regurgitation with systolic anterior movement (SAM) of the anterior mitral leaflet (Figure 2A, see
supplementary movie file). The left ventricular ejection fraction was decreased to 53.0%, as evaluated
by the biplane method of disks. Serum CK, CK-MB, and Troponin T were mildly elevated to 210 U/L,
21 U/L, and 0.447ng/mL, respectively. Emergent cardiac catheterization was performed to eliminate
the possibility of acute coronary syndrome. Coronary artery stenosis and occlusion were not found
on coronary angiography. Left ventriculography confirmed apical akinesis, circumferential severe
hypokinesis of the mid ventricle, basal hyperkinesis, and severe mitral regurgitation (Figure 2B).
The intra-left ventricular pressure gradient was approximately 50 mmHg by catheterization. Based
on these findings, the patient was diagnosed with Takotsubo cardiomyopathy.
The patient had been diagnosed with ventricular septal hypertrophy with LVOTO (pressure
gradient = 14 mmHg) and sick sinus syndrome by her family doctor one month previously. She was
administered 100mg cilostazol per day from October 25 to treat symptomatic sinus bradycardia,
and the dose was increased to 200mg per day from October 26. Takotsubo cardiomyopathy was
presumed to have developed suddenly following the dose increase in cilostazol because neither
electrocardiogram nor echocardiography exhibited an ST-T abnormality or apical hypokinesis,
respectively, on October 26 (Figure 1B). She was hospitalized following cardiac catheterization.Fukata M, et al
Cilostazol was discontinued from the date of admission. The
administration of cibenzoline was initiated to ameliorate LVOTO.
She developed acute pulmonary edema and was treated with diuretics
and human atrial natriuretic peptide. A permanent pacemaker was
implanted for the treatment of excessive bradycardia. Her symptoms
were gradually improved along with the amelioration of the left
ventricular wall motion abnormality. Neither LVOTO nor SAM
could be detected on echocardiography at discharge. Serum BNP
level dropped from 1047.2pg/mL on admission to 139.1pg/mL before
discharge.
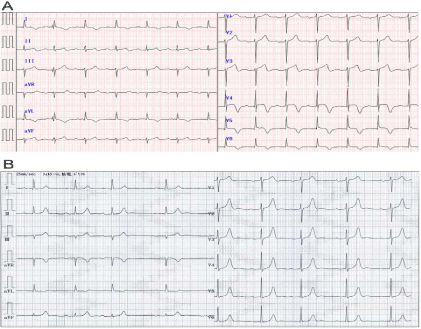
Figure 1
Figure 1
An electrocardiogram on admission (A) exhibited an elevated ST segment in leads II, III, aVF, and V2-5 and negative T wave in leads I, aVL, and V4-6.
No ST-T abnormality was present in an electrocardiogram 2 days before admission (B).
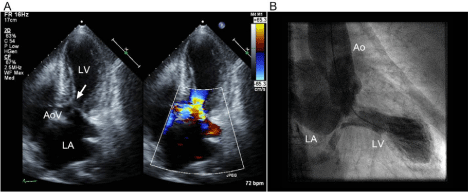
Figure 2
Figure 2
Apical 5 chamber view of echocardiographic image at systole (A) showed left ventricular outflow tract obstruction (LVOTO) with acceleration flow
and systolic anterior movement (SAM, indicated by an arrow) of the anterior mitral leaflet with severe mitral regurgitation. Left ventriculography at end-systole
(B) demonstrated apical ballooning and basal hyper-contraction. The left ventricular chamber adopted a characteristic configuration that mimics Takotsubo.
Abbreviations: LA, left atrium; LV, left ventricle; AoV, aortic valve; Ao, ascending aorta. Wall motion abnormality and dynamic obstruction in LV is shown in the
movie (supplementary file).
Discussion
Takotsubo cardiomyopathy is a well-described disease entity that
is characterized by transient apical or mid-ventricular left ventricular
dysfunction in response to sudden, unexpected emotional distress and
is relatively common in elderly woman. Enhanced cardiomyocytes
stimulation by catecholamines related to emotional or physical
distress is considered to play a major role in the pathogenesis of
this cardiomyopathy from the findings in the human study and the
experiment using the animal model [4,5]. Catecholamine-induced
myocardial stunning in Takotsubo cardiomyopathy is possibly mediated by a switch in β2 adrenoceptor coupling from Gs protein signaling to Gi protein signaling [6]. Since it had been demonstrated
that intracellular cyclic adenosine monophosphate (cAMP)-
dependent protein kinase A phosphorylation of the β2
adrenoceptor initiate this switch in signal trafficking [7], cAMP increases in the
ventricular myocardium can exert not only positive inotropism in the
base via β1 adrenoceptor-Gs signaling, but also negative inotropism in
the apex via β2 adrenoceptor-Gi signaling [6]. Additionally, negative
inotropism by catecholamines is possibly induced by cAMP-mediated
calcium overload [8]. Therefore, cilostazol, a selective inhibitor of
phosphodiesterase III, could cause Takotsubo cardiomyopathy by
suppressing cAMP degradation with the resultant accumulation of
intracellular cAMP. In addition to neurohumoral factors, an intraleft
ventricular dynamic gradient, as was observed in this case,
was previously shown to be a prerequisite for the development of
Takotsubo cardiomyopathy [9]. Since this patient had no episode
of significant mental distress before the onset of chest discomfort,
the pharmacological action of cilostazol to increase cAMP in a heart
possessing a structural prerequisite could have likely precipitated
Takotsubo cardiomyopathy.
To our best knowledge, this is the first report that cilostazol could induce Takotsubo cardiomyopathy. Most of the drugs that have
been reported as a possible trigger of Takotsubo cardiomyopathy
are considered to precipitate Takotsubo cardiomyopathy by
catecholamine surge or by overstimulation of the sympathetic
nerve system [3]. We suppose that in addition to these sympathetic
agonists or sympathomimetic drugs, the drugs that upregulate
the molecules mediating adrenoceptor signaling could cause this
cardiomyopathy. Supporting this supposition, there are three case
reports of Takotsubo cardiomyopathy induced by the administration
of the cAMP increasing drugs (levothyroxine [10], anagrelide
[11], dipyridamole [12]). The pathophysiological role of cAMP in
Takotsubo cardiomyopathy should be specifically examined in future
to further understand the disease.
This study describes an informative case in which cilostazol,
a commonly used drug in the cardiovascular field, could have
triggered Takotsubo cardiomyopathy. This case highlights the
role of intracellular cAMP levels in the pathogenesis of Takotsubo
cardiomyopathy and suggests the need for careful observation
following the administration of a phosphodiesterase inhibitor as
well as other cAMP-increasing agents, especially to a patient with an
intra-left ventricular obstruction.
References
- Sharkey SW, Windenburg DC, Lesser JR, et al. Natural history and expansive clinical profile of stress (tako-tsubo) cardiomyopathy. J Am Coll Cardiol. 2010; 55: 333-341.
- Eitel I, von Knobelsdorff-Brenkenhoff F, Bernhardt P, et al. Clinical characteristics and cardiovascular magnetic resonance findings in stress (takotsubo) cardiomyopathy. JAMA. 2011; 306: 277-286.
- Amariles P. A comprehensive literature search: drugs as possible triggers of Takotsubo cardiomyopathy. Curr Clin Pharmacol. 2011; 6: 1-11.
- Wittstein IS, Thiemann DR, Lima JA, Baughman KL, Schulman SP, Gerstenblith G, et al. Neurohumoral features of myocardial stunning due to sudden emotional stress. N Engl J Med. 2005; 352: 539-548.
- Ueyama T, Kasamatsu K, Hano T, Yamamoto K, Tsuruo Y, Nishio I. Emotional stress induces transient left ventricular hypocontraction in the rat via activation of cardiac adrenoceptors: a possible animal model of 'tako-tsubo' cardiomyopathy. Circ J. 2002; 66: 712-713.
- Lyon AR, Rees PS, Prasad S, Poole-Wilson PA, Harding SE. Stress (Takotsubo) cardiomyopathy - a novel pathophysiological hypothesis to explain catecholamine-induced acute myocardial stunning. Nat Clin Pract Cardiovasc Med. 2008; 5: 22-29.
- Daaka Y, Luttrell LM, Lefkowitz RJ. Switching of the coupling of the beta2-adrenergic receptor to different G proteins by protein kinase A. Nature. 1997; 390: 88-91.
- Mann DL, Kent RL, Parsons B, Cooper G 4th. Adrenergic effects on the biology of the adult mammalian cardiocyte. Circulation. 1992; 85: 790-804.
- Merli E, Sutcliffe S, Gori M, Sutherland GG. Tako-Tsubo cardiomyopathy: new insights into the possible underlying pathophysiology. Eur J Echocardiogr. 2006; 7: 53-61.
- Kwon SA, Yang JH, Kim MK, Park SW, Kim SH, Park KH, et al. A case of Takotsubo cardiomyopathy in a patient with iatrogenic thyrotoxicosis. Int J Cardiol. 2010; 145: e111-113.
- Proietti R, Rognoni A, Ardizzone F, Maccio S, Santagostino A, Rognoni G. Atypical Takotsubo syndrome during anagrelide therapy. J Cardiovasc Med (Hagerstown). 2009; 10: 546-549.
- Koh AS, Kok H, Chua T, Keng F. Takotsubo cardiomyopathy following dipyridamole pharmacologic stress. Ann Nucl Med. 2010; 24: 497-500.